Ecotoxicological and biotechnological characterization of biologically active substances from the marine environment
Within sub-activity 2.3. in the period from 2023-2025. year we continued with basic research aimed at elucidating the repair mechanism of specific DNA damage (DNA-protein crosslinks – DPCs) and the role of targeted membrane transport proteins in protection against pollutants from the aquatic environment, mostly using the zebrafish (Danio rerio) as a research model. We have obtained significant new findings related to the role of the enzyme tyrosyl-DNA phosphodiesterase 2 (Tdp2) in DPC repair (Fig. 1), as well as in unraveling the interactions of relevant pollutants of the aquatic environment with the so-called Mate3 (Fig. 2), Oap1d1 (Fig. 3) and Abcg2a (Fig. 4) zebrafish membrane transporters. These findings bring us closer to the development and affirmation of new ecotoxicological biomarkers of water pollution.
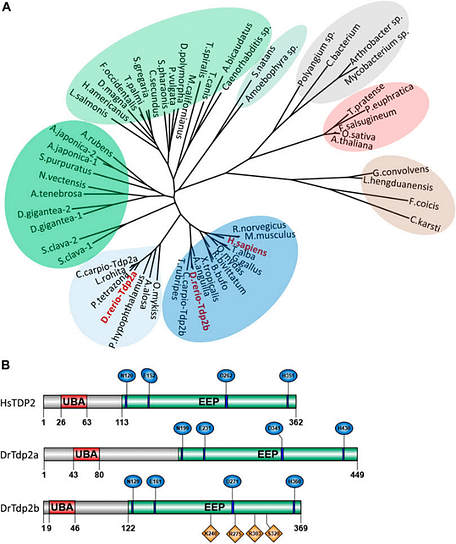
Figure 1. Phylogenetic analysis and domain organization of tyrosyl-DNA phosphodiesterase 2 in humans and zebrafish. (A) Phylogenetic tree of tyrosyl-DNA phosphodiesterase 2 (TDP2). (B) Domain structures of human and zebrafish tyrosyl-DNA phosphodiesterase 2. From: Antičević et al., 2024, Front. Cell Develop. Biol.
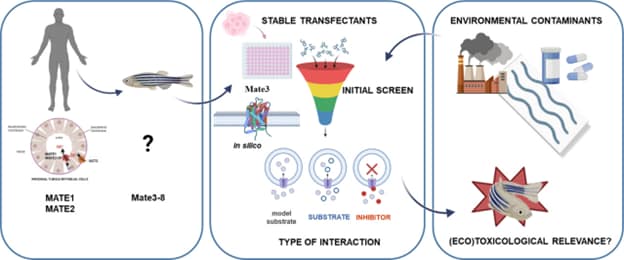
Figure 2. Summary of the research sequence aimed at identifying environmental pollutants that inhibit the transport activity of the zebrafish Mate3 protein. From: Vujica et al., 2023, Sci Total. Environ.
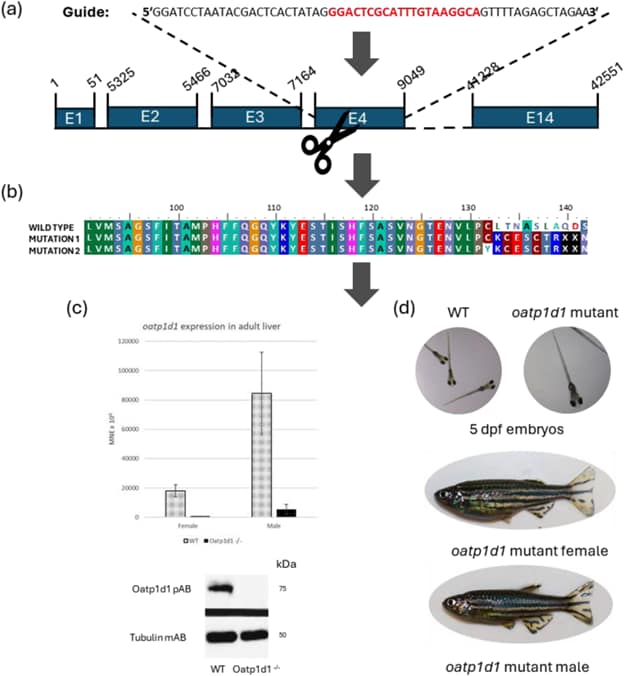
Figure 3. Development of genetically modified zebrafish to investigate the role of the Oatp1d1 transporter. (a) Schematic representation of the CRISPR/Cas9 guide and targeting of exon 4 of the oatp1d1 gene; (b) Sequences of Oatp1d1 protein and mutants with a reading frame shift causing a stop codon at position 140; (c) RT-qPCR results showing reduced oatp1d1 transcript expression in the liver of adult oatp1d1 mutants; (d) Images of mutant and wild-type embryos. From: Vujica et al., 2024, Aquat. Toxicol.
A)
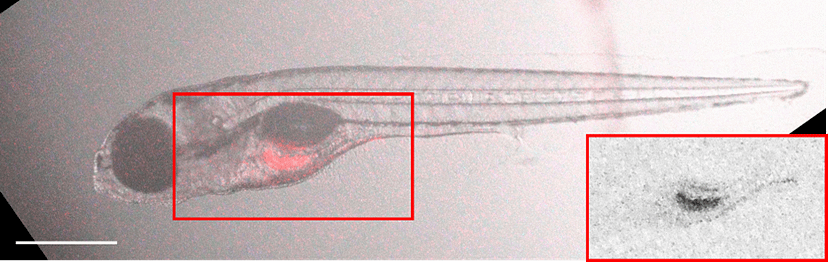
B)
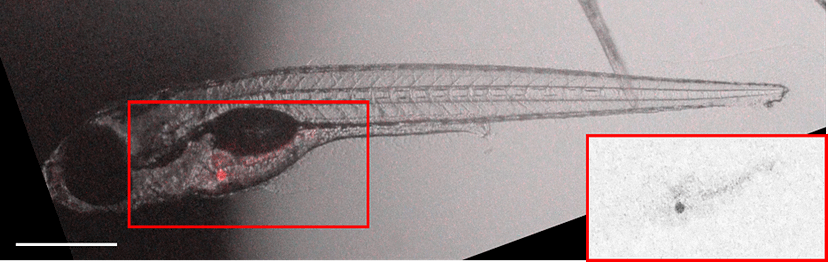
Figure 4. Accumulation pattern of the model pollutant pheophorbide A (red staining) in live, anesthetized wild-type (A) and Abcg2a transporter-deficient zebrafish larvae (B) 5 days after treatment. From: Lončar et al., 2026, Aquat. Toxicol.
